Stoichiometric Defects – The compounds which obey the law of definite proportions, the law of constant composition and the law of conservation of mass are called stoichiometric compounds. Point defects can be further divided into following types – These may be larger or smaller than the normal atoms in the lattice. Impurity atoms(Interstitial or Substitutional Type)Īn impure atom is present on the lattice by substituting a lattice site atom then it is known to be a substitutional impurity atom. When an atom is forced into an interstitial position from a normal lattice site by creating a vacancy, the resulting pair (which is a vacancy and the interstitially together) is known as a Frenkel Defect.Ĥ. 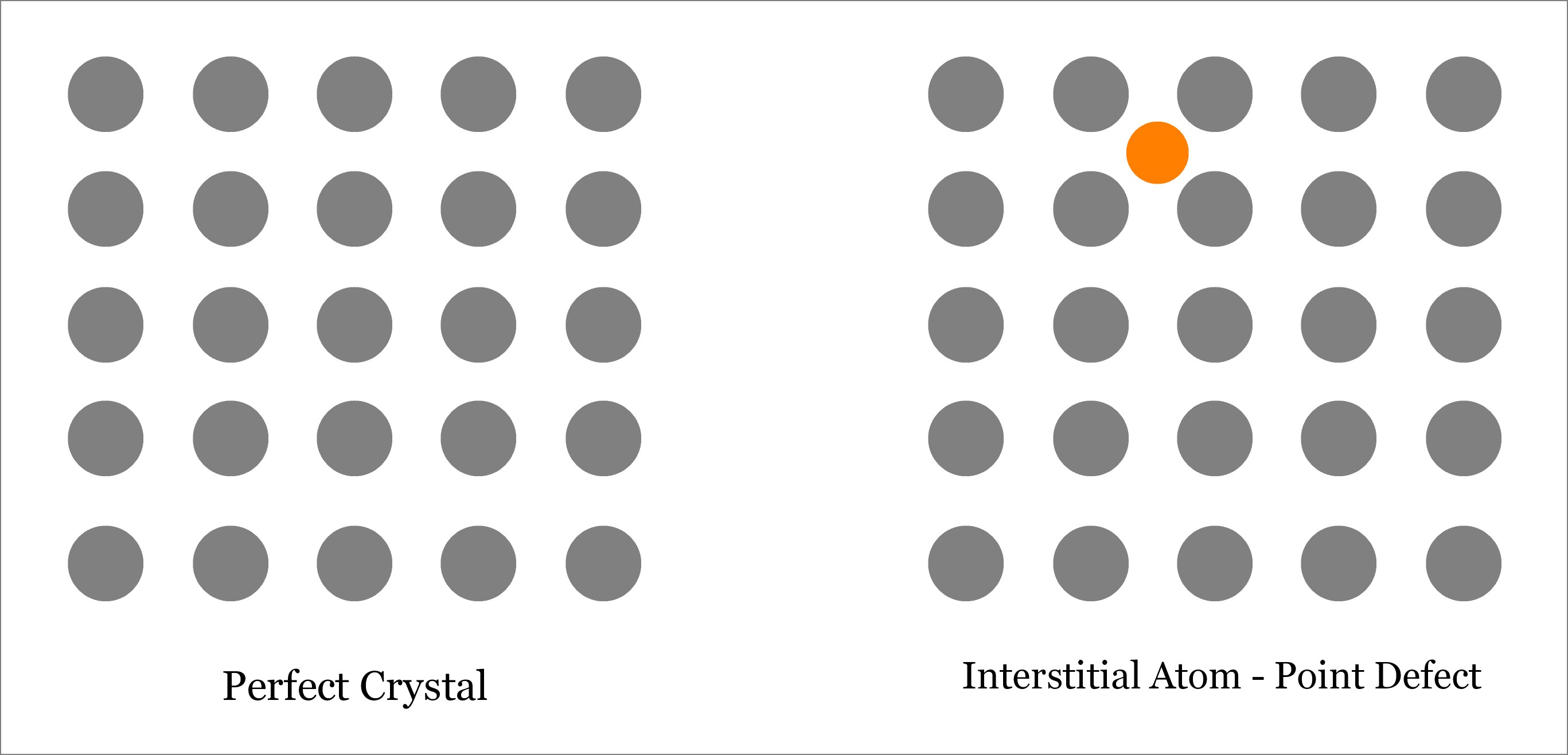
As compared to the number of vacancies, the number of interstitials is negligibly small in an ordinary metal. Generally, the size of the interstitial is smaller than a vacancy. When an atom occupies an interstitial site of the metal, it is known as Interstitial. They can be di-vacancies or tri- vacancies. In a Crystal structure of metal when an atom misses from its lattice site, it is known as a vacancy or vacant lattice site. Latest discuss some of the types of point defects It only extends a few diameters beyond its lattice position. The size of the defect could be one or two atomic diameters which are just like a point. The frequently asked questions at the end of the article can help you with your doubts if you incur any while reading the same.Ī Localized disruption in the regularity of a lattice is known as a point effect. This article covers the meaning and the various types of point defects. Original atom of the crystal is replaced by foreign atom. One or more atoms of the crystal are missing from their corresponding lattice site.Ītom/s is shifted from its corresponding lattice site to interstitial position in the crystal.įoreign atoms occupy the interstitial position in the crystal lattice. These are the smallest possible defects in any crystalline solid material. That’s why they are also called zero dimensional (0-D) defects. They do not extend in space in any dimension. These defects occur only at or around a single lattice point. Those defects in the crystals which occur around an atom or particle are called point defects. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
